What Is Bio-Coordination Chemistry, and How Does It Relate to Metalloenzymes?
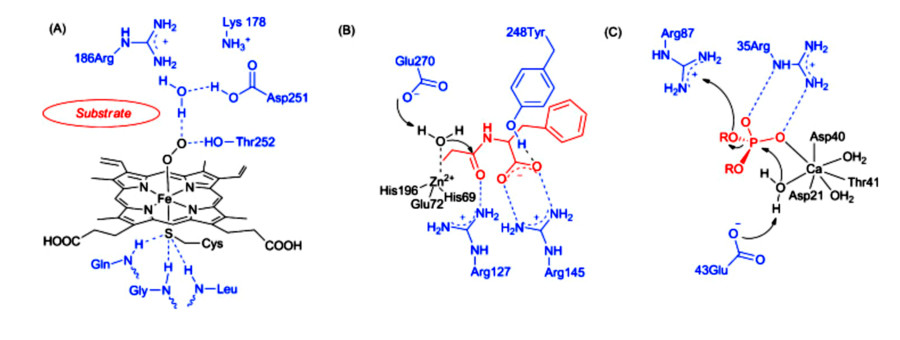
Bio-coordination chemistry is a subset of inorganic chemistry and biology concerned with metal-ligand interactions. Metalloenzymes—natural coordination compounds formed by metal ions and protein scaffolds—represent a core focus of this field. Metalloenzymes are ubiquitous and important for physiological and catalytic regulation in microbial, plant, and mammalian cells. Hydrogenases and nitrogenases are examples of enzymes with complex metal-ligand frameworks. These metalloenzymes can directly catalyze useful chemical transformations, such as the energy-efficient production of hydrogen gas and the conversion of atmospheric nitrogen to ammonia under ambient conditions, in the presence of various other substrates.
Natural metalloenzymes are often complex in structure and sensitive to various environmental stressors, making their isolation and large-scale production challenging. Therefore, it is of great significance to study their structure-activity relationships and design metalloenzyme mimics with low synthesis costs and high stability.
Theoretical Basis and Design Strategies for Mimicking Enzyme Catalysis
Metalloenzyme mimicry is designed by mimicking the structural and electronic features of natural enzyme active sites. This involves selecting appropriate metal ions (e.g., Fe, Zn, Cu, Mn) and engineering their coordination environments using macrocyclic ligands, nucleic acids, or supramolecular hosts such as cyclodextrins, crown ethers, calixarenes, and porphyrins. These systems offer tunable geometry, redox potential, and ligand field strength—key parameters that govern catalytic performance.
Modern design strategies include:
a. Structural Mimicry: Reconstructing the spatial arrangement of native metalloenzyme centers to emulate substrate binding and transition state stabilization.
b. Microenvironment Engineering: Embedding active sites in supramolecular scaffolds to control local polarity, pH, and steric hindrance.
c. Computational Modelling: Employing DFT and molecular dynamics to predict reactivity and guide ligand design.
d. Multi-functionality: Incorporating redox-active or photo-responsive components for enhanced regulation.
A prominent example is the development of a Pt-Au alloy catalyst that mimics the active site of formate oxidase, enabling high-yield aerobic oxidation of formate to hydrogen peroxide under ambient conditions. This catalyst exhibits H2O2 yields far exceeding those of electrocatalysts and photocatalysts, and its catalytic efficiency is significantly superior to that of the natural enzyme.
What Are the Practical Applications of Metalloenzyme Catalysis?
Green Chemistry and Sustainable Energy
Artificial metalloenzymes are transforming oxidative and reductive catalysis in green chemistry. Systems based on cobalt, iron, or manganese mimic peroxidases, oxidases, and hydrogenases, enabling selective transformations under mild conditions without hazardous reagents. For instance:
| Application Area | Example Catalyst System | Function |
| H2 Production | [FeFe]-Hydrogenase Mimics | Catalytic hydrogen evolution |
| H2O2 Synthesis | Pt-Au Alloy Complexes | Aerobic formic acid oxidation |
| Oxidative Biotransformation | Metalloporphyrins, Cu-Zn mimics | Hydrocarbon hydroxylation |
These technologies not only reduce environmental impact but also offer scalability for industrial use. Alfa Chemistry supports innovation in this space through custom synthesis services for porphyrins, macrocyclic ligands, and tailored coordination compounds.
Biomedicine and Drug Discovery
Metalloenzymes are vital targets and tools in modern medicine. Zinc-dependent enzymes such as carbonic anhydrase and matrix metalloproteinases (MMPs) are involved in processes ranging from pH regulation to tumor invasion. Inhibitors targeting these enzymes serve as therapeutics for cancer, neurodegenerative disorders, and infectious diseases.
Furthermore, synthetic metalloenzymes offer a platform for developing new biomaterials with catalytic, imaging, or therapeutic functions. Single-atom nanozymes (e.g., Co3O4-Zn coatings) display tunable oxidase-like activity and are being explored for wound healing, antimicrobial coatings, and ROS generation in cancer therapy. For more updates on biotechnology and healthcare innovations, you may also visit the Health section of News Times Wire.
Environmental Remediation
Mimetic systems of superoxide dismutase (SOD) and peroxidase have been implemented in pollutant degradation, heavy metal detoxification, and soil remediation. Due to their robustness and low cost, artificial metalloenzymes surpass their natural counterparts in terms of field deployability. With enhanced redox cycling and pollutant binding capacities, these catalysts serve in advanced oxidation processes (AOPs) for water treatment and environmental decontamination.
How Does Alfa Chemistry Contribute to Advancing This Field?
Alfa Chemistry offers metal salts, cyclodextrins, crown ethers, calixarenes, porphyrins, nucleotides, and their derivatives to aid in metalloenzyme simulation research! Our products are of high purity and are available from stock with stable batch quality!
What Challenges Remain in Metalloenzyme Research?
Although they hold significant promise, artificial metalloenzymes also encounter ongoing challenges:
- Catalytic Specificity: In contrast to their native counterparts, most synthetic artificial metalloenzymes possess a broad substrate scope, which often leads to undesired side reactions.
- Structural Complexity: Mimicking the structure of multi-metallic clusters and capturing transient intermediate states are synthetically challenging.
- Scalability: High-yield and cost-effective synthesis of stable and robust enzyme mimics remains a major hurdle in their commercial applications.
- Stability: Many systems are sensitive to degradation under operational conditions, such as oxidation or photodecomposition.
Future research will focus on areas such as modular ligand design, integration with protein engineering, and the development of biosensors based on metal-ligand recognition for diagnostic applications.
FAQs about Metalloenzyme Mimics
1. Can synthetic metalloenzymes match the selectivity of natural enzymes?
While current systems offer promising activity, achieving the same substrate specificity as native enzymes remains a key challenge. Research is ongoing into microenvironment design and artificial active sites.
2. What types of ligands are most commonly used in enzyme mimicry?
Porphyrins, macrocyclic compounds (e.g., crown ethers and calixarenes), and nitrogen-donor ligands such as imidazoles and pyridines are widely used due to their coordination versatility and biomimetic properties.
3. Are there commercial applications of artificial metalloenzymes?
Yes, industrial examples include oxidation of hydrocarbons, green H2O2 production, and enzymatic fuel cells. They are also being evaluated in drug screening and biosensing platforms.
4. How stable are nanozymes under physiological conditions?
Stability varies by formulation, but many nanozymes exhibit robust activity across a wide pH range and temperature spectrum, particularly when stabilized by polymeric or inorganic coatings.
5. Can metalloenzymes be used in vivo for therapeutic purposes?
Yes, especially nanozyme-based systems with ROS-generating capabilities, which are being studied for applications in tumor suppression, antimicrobial therapy, and neuroprotection.

